📌 Key Takeaways
A single approved sample proves a factory can build it once—not that they can build it the same way every time.
- Approval Is the Starting Gate, Not the Finish Line: Signing off on one perfect unit confirms the factory’s capability, not their ability to repeat it across thousands of units.
- Add “Limit Samples” to Define the Edges: High-side and low-side boundary samples show exactly how far production can drift before a unit becomes unacceptable.
- Replace Memory-Based QC with Measured Limits: When technicians compare units to memory instead of recorded data, “sounds close enough” becomes the real—and unreliable—standard.
- Store Sealed Copies in Two Locations: Keep one matched set at the factory and one at your headquarters so disputes can be resolved with evidence, not arguments.
- Re-Validate After Any Change: Component swaps, supplier switches, or production gaps all require new approval before shipping resumes.
A sample on a shelf is not a quality system—it’s just the foundation for one.
Product teams managing private-label amplifier programs will gain a practical framework for enforcing consistency, preparing them for the detailed checklist that follows.
~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~
The first production shipment arrives. Your QA team unboxes a unit, powers it up, and runs a frequency sweep. The numbers don’t match. The midrange sounds flatter than the sample sitting in your reference library. You call the factory. They claim the unit is “within tolerance.”
But tolerance to what?
You approved one unit. They built three thousand. And now your team is arguing about whether this is acceptable drift or a contract violation—while pallets sit in receiving and your launch date doesn’t move.
Within an hour, you’re in a familiar cross-functional standoff. Product wants to protect the launch date. QA wants a stop-ship until the root cause is clear. Procurement wants leverage before the supplier locks into “it’s within tolerance.” Support is already picturing the customer complaints and brand reputation damage that follow a flawed shipment.
Here’s the hard truth: the argument usually isn’t about sound. It’s about authority. Nobody can prove what “matches the sample” actually means.
This is the Golden Sample trap. Your approved sample proved the factory can build it once. It didn’t prove they can build it every time.
A Golden Sample is the mutually approved physical reference unit that defines what “correct” looks and measures like for production. While your controlled documentation—such as your PLM data, Gerber files, and BOM—explicitly locks the technical specifications, the Golden Sample serves to validate them. It provides a shared physical benchmark. But on its own, it proves baseline capability—not long-term consistency. To turn that single reference into a manufacturing firewall against quality drift, you need more than a trophy unit in a cabinet. You need a documented system: the Golden Sample paired with Limit Samples, objective QC limits, and dual-side governance that makes “match the sample” measurable, auditable, and enforceable.
This problem shows up most often in private-label amplifier programs because “close enough” can’t be judged by a quick visual inspection. If you’re qualifying amplifier manufacturers, you need a production control system that turns “match the sample” into something your QA team can actually enforce.
If you’re shopping for a cheap workaround or DIY approach, this framework isn’t for you. This is about preventing brand-damaging quality drift in mass production.
Myth: One Perfect Golden Sample Means the Factory ‘Can Do It’
What Is a Golden Sample in Manufacturing?
A Golden Sample is the approved reference unit that defines exactly what you ordered: the BOM revision, firmware version, acoustic signature, cosmetic finish, and packaging spec. When both parties sign off, that unit becomes the contract anchor—the physical embodiment of “build it like this.”
The problem isn’t the Golden Sample itself. The problem is treating approval as the finish line rather than the starting gate.
Signing off on a Golden Sample confirms capability. The factory demonstrated they could assemble the correct components, tune the DSP correctly, and pass your listening test. Once. Under controlled conditions. Often with their best technicians watching.
That single unit tells you nothing about what happens at unit 500, unit 2,000, or after the second component substitution. It doesn’t define how far production can drift before crossing from “acceptable variation” into “defect.” And it doesn’t give your QA team objective criteria to enforce during a dispute.
The decision point you need to make is early: are you approving a unit, or are you approving a repeatable production definition?
A practical proof artifact keeps that decision from becoming philosophical. Before you treat approval as a gate, require three items in writing: the factory’s production test plan that will be used at end-of-line (inputs, limits, pass/fail rules), the baseline measurement set tied to the Golden Sample (curves, not just a listening note), and the change-control rule that defines what forces re-validation. If those artifacts don’t exist, the Golden Sample isn’t a control point. It’s a memory.
For a deeper sign-off workflow that turns approval into a documented gate, use the Golden Sample approval checklist.
Reality: Capability Is Not Consistency
Why Do Production Units Sound Different from the Sample?
Production units sound different from the sample when variation enters the build without a measured boundary. That variation can come from component lots, substitutions, process changes, firmware changes, or inconsistent interpretation of “acceptable.” If your acceptance method relies mostly on subjective listening, the argument becomes unresolvable.
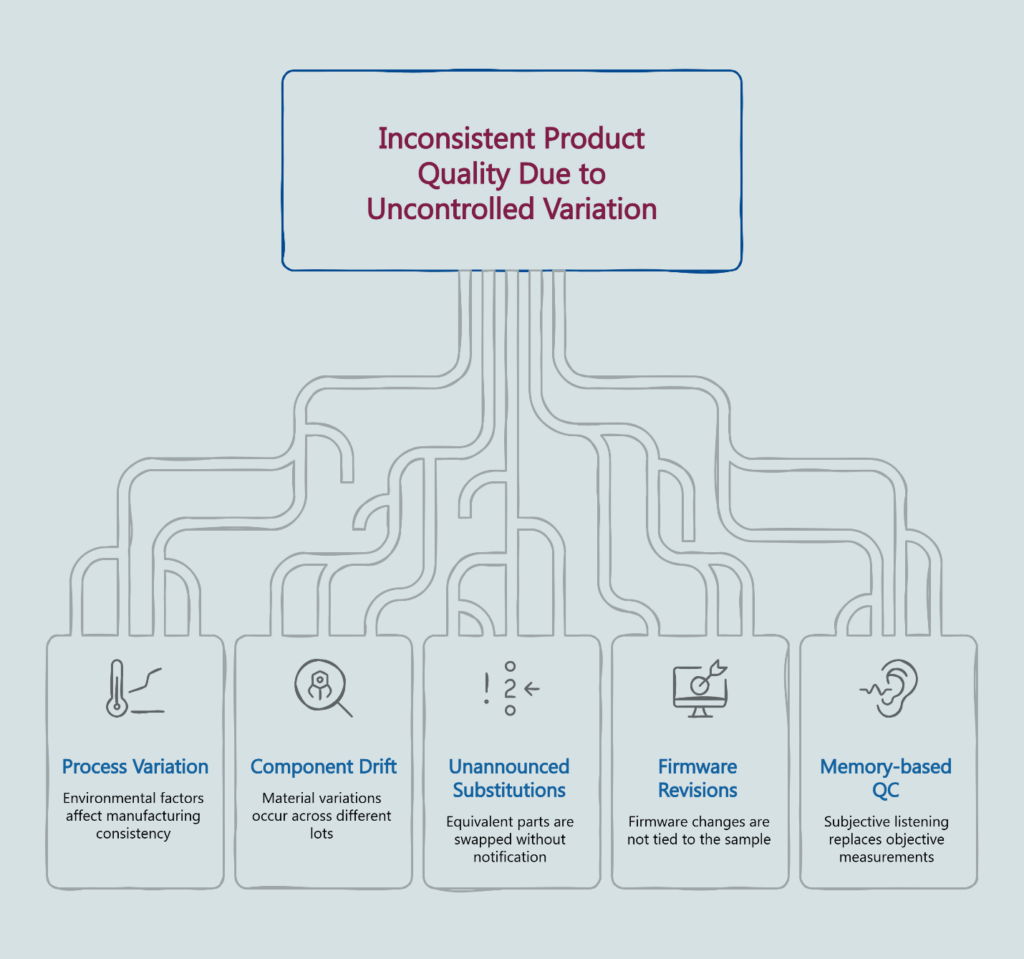
Many product teams generally assume that quality drift will manifest as an obvious, catastrophic failure. In practice, drift is typically a gradual statistical degradation that technically remains within the broad spec sheet while audibly moving away from your Golden Sample.
Process variation is inherent in any manufacturing environment. Ambient temperature affects adhesive cure times. Humidity shifts change material properties. Even well-documented processes produce units that cluster around a target—not units that hit it identically.
Component drift occurs naturally across material lots. The voice coil wire from this month’s supplier shipment may test within spec but behave differently than last month’s. Capacitors age. Magnets vary.
Unannounced substitutions happen when a component goes on allocation or a supplier changes. A factory operating under cost pressure may swap to an “equivalent” part without formal notification—especially if your Engineering Change Order (ECO) process isn’t airtight. Procurement sees “equivalent.” Your customers hear “different.”
Firmware or DSP revisions that aren’t bound to the sample create mixed behavior. If the Golden Sample isn’t explicitly tied to a firmware/DSP revision, you can end up shipping mixed behavior under one SKU.
Memory-based QC compounds the problem. When end-of-line testing relies on technicians listening and comparing to memory rather than measuring against documented limits, “sounds close enough” becomes the de facto standard.
The result? Units that pass subjective checks but fail when measured. Or units that measure differently but nobody catches at end-of-line—shipping variance that should have been contained at the factory.
The real cost isn’t only the defective units. It’s the organizational drag. Every ambiguity forces a consensus-based organization to do what it always does under risk: escalate, argue, and stall. That drag costs schedule, margin, and reputation long before a shipment leaves the dock.
If you’ve lived through this once, the adjacent pattern will feel familiar: drift often appears after the first run, when the program moves from “special attention” to routine execution. That’s the core risk described in quality drift after the first run.
The Fix: Add a ‘Limit Sample’ and Objective Limits
How Do I Ensure the Factory Matches the Sample Exactly?
You don’t—not exactly. What you ensure is that production stays within defined boundaries that protect the customer experience.
Limit Samples define the edges of acceptable variation. If your Golden Sample represents the target, Limit Samples represent “as far as you can drift in each direction and still be acceptable.” One shows the high-side boundary; another shows the low-side boundary. These aren’t failures—they’re calibrated references that answer the question every QA team asks: “How much is too much?”
Objective baselines convert your Golden Sample’s performance into measurable pass/fail criteria. Systems like KLIPPEL QC capture frequency response curves, impedance profiles, and distortion measurements from your approved unit. Production units then test against those curves—not against a technician’s recollection. The machine doesn’t have a bad day or rush before lunch. It compares data to data.
When you combine Golden Sample + Limit Samples + objective test limits, you build what we call a manufacturing firewall. It detects and strictly limits quality drift before it becomes an unresolvable dispute. It makes “within tolerance” mean something specific, measurable, and enforceable.
If you want a quality system language that aligns stakeholders and auditors, anchor this approach inside a QMS mindset. ISO 9001:2015 provides an international standard for quality management systems and a structure for delivering consistent products. ASQ’s summary reinforces the same point: consistency comes from system design, not hope.
The proof artifact to request here is straightforward: the limit file and the calibration rule. If the supplier can’t show you how limits are calculated, stored by revision, and protected by change control, you’re still operating on trust and memory.
Golden Sample Governance: Signing, Sealing, and Dual-Side Storage
How Should Golden Samples Be Stored and Controlled?
A Golden Sample without custody control is a future argument waiting to happen.
The scenarios are predictable: Factory claims they built to spec. Brand claims they didn’t. Both reference “the Golden Sample.” Except the factory’s sample has been on the production floor for eight months, handled by multiple technicians, and nobody’s sure if it’s the original or a replacement. Meanwhile, the brand’s sample is in a different office than the QA team and hasn’t been measured since approval.
Proper governance prevents this. Here’s the protocol:
Seal and uniquely label each unit in the Golden Sample set (including Limit Samples). Use tamper-evident packaging. Assign serial numbers or unique IDs that appear in the sign-off documentation.
Create a dual sign-off sheet capturing: date, location, sample IDs, BOM revision, firmware/DSP revision, cosmetic/packaging revision, Brand signatory (typically Product/QA Owner), and Factory signatory (QA Manager or Production Manager).
Store matching copies in two locations: one at the factory QC station, one at brand HQ or a designated testing lab. Both sets must be identical and controlled. If the factory needs to reference the sample, it’s in their facility. If your team needs to verify a complaint, you have a clean reference.
Log access and custody. When someone opens the sample, it’s recorded. This isn’t paranoia—it’s the audit trail that resolves disputes in minutes rather than weeks.
Common Failure Modes and How to Prevent Them
What Should Trigger a Re-Approval of the Sample Set?
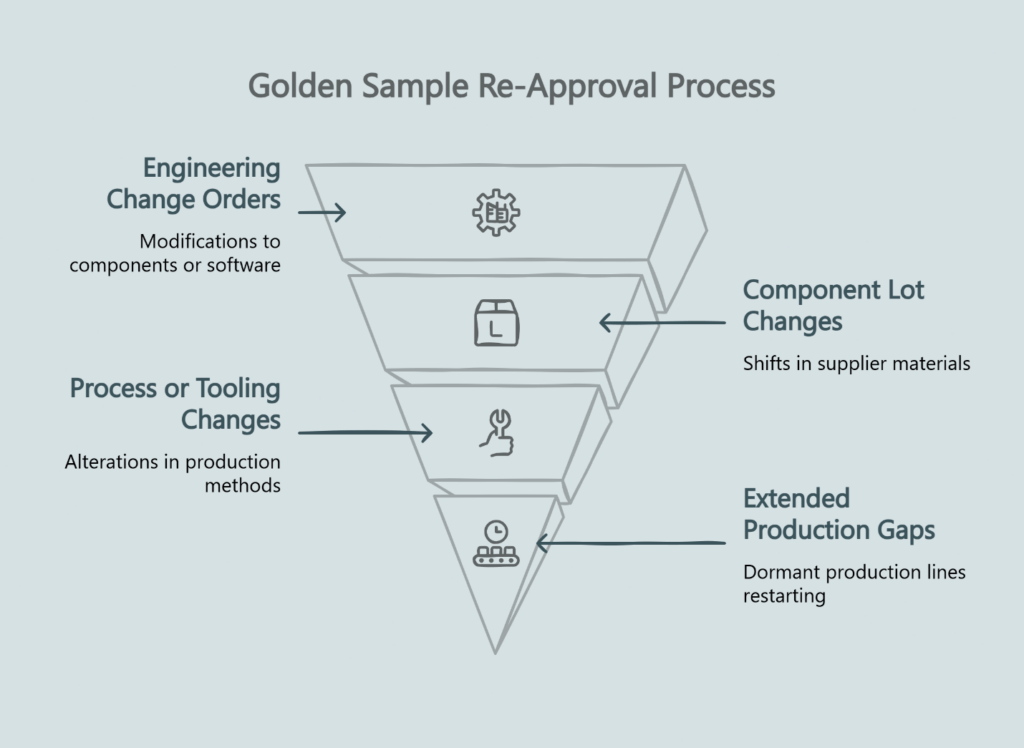
Treat any change that could affect performance as a trigger for re-validation:
Engineering Change Orders (ECOs) that modify components, materials, or software require new Golden Sample approval. Even “equivalent” substitutions can shift acoustic performance outside your limits. For deeper coverage on change management, see our guide on Engineering Change Control for OEM programs.
Component lot changes from your supplier’s supplier matter. If the factory switches to a new voice coil wire supplier—even if it meets the same published specs—your QC limits may need adjustment.
Process or tooling changes affect consistency. New adhesive dispensing equipment, different curing ovens, or revised assembly sequences all warrant verification against the baseline.
Extended production gaps create risk. If a product line goes dormant for six months and then restarts, re-validate before the first shipment.
The discipline here connects directly to traceability. When your factory maintains barcode or QR systems linking unit serial numbers to component lots and test data, re-validation triggers become manageable. Without traceability, you’re guessing which units might be affected.
The upstream prevention move is also worth stating: late changes are expensive because they force re-validation under time pressure. A disciplined DFM review before tooling reduces the number of “surprise” changes that show up during ramp.
Golden Sample Protocol Checklist
Use this checklist to convert a single approved unit into an enforceable production standard.
1. Define what the Golden Sample covers: BOM revision, firmware/DSP revision, cosmetic finish and packaging, test limits (frequency response, distortion, impedance).
2. Build the Golden Sample under production conditions: Use intended production tooling and processes—not a one-off lab build. Document build conditions (date, line, technicians).
3. Capture objective baselines: Record frequency response, distortion, and impedance curves. Store data in a format your QC equipment can reference.
4. Define and label Limit Samples: High-side boundary (acceptable but near the upper tolerance edge) and low-side boundary (acceptable but near the lower tolerance edge).
5. Translate baselines into acceptance limits: Establish pass/fail criteria tied to baseline curves. Specify which parameters are critical vs. informational.
6. Create dual sign-off documentation: Date, location, sample IDs/serials, BOM revision, firmware revision, cosmetic revision IDs. Brand signatory (role): Product/QA Owner. Factory signatory (role): QA Manager / Production Manager.
7. Seal and label the sample set: Tamper-evident packaging with unique serial numbers matching sign-off sheet.
8. Store matching copies in two locations: Factory (QC station or designated controlled storage) and Brand HQ (designated lab or secure storage).
9. Train production/QC teams: Objective test first (instrument measurement), subjective spot-check second (listening verification).
10. Re-validate after any change: ECO / component substitution, process shift or tooling change, extended production gap.
For a detailed walkthrough of the sign-off process, see the Golden Sample approval checklist and the practical workflow in our guide on how Golden Sample QA prevents defective inventory.
Conclusion: Treat Golden Samples as a System, Not a Trophy
The Golden Sample trap catches brands that confuse approval with assurance. While a singular unit demonstrates factory capability, long-term consistency requires strict adherence to this documented triad of limits, reference units, and bilateral governance.
That system establishes a rigid quality gate. It answers the dispute before it happens. It gives your QA team enforceable criteria. And it protects your brand from the kind of quality drift that turns a successful launch into a reputation problem.
The sample on the shelf isn’t the finish line. It’s the foundation for everything that follows.
The next time a supplier says “within tolerance,” you won’t need a debate. You’ll need the revision ID, the baseline curve, and the pass/fail result. If you want to pressure-test your current sample governance before the next ramp, contact our team to talk through the control plan and the evidence you should require before committing to volume.
Disclaimer: This content is for informational purposes and should not replace professional manufacturing or quality assurance advice specific to your program requirements.
Our Editorial Process:
Our expert team uses AI tools to help organize and structure our initial drafts. Every piece is then extensively rewritten, fact-checked, and enriched with first-hand insights and experiences by expert humans on our Insights Team to ensure accuracy and clarity.
About the China Future Sound Insights Team
The China Future Sound Insights Team is our dedicated engine for synthesizing complex topics into clear, helpful guides. While our content is thoroughly reviewed for clarity and accuracy, it is for informational purposes and should not replace professional advice.






