📌 Key Takeaways
A factory’s quality paperwork can look perfect while actual production quietly drifts out of spec under deadline pressure.
- Systems Beat Checklists: Quality gates only work when a digital system — not a person — blocks untested units from moving forward.
- Golden Samples Prove Capability, Not Consistency: That approved sample was built with extra care; it doesn’t prove thousands of units will match it under time pressure.
- One Skipped Check Triggers a Chain Reaction: A single bypassed test station can lead to warranty claims, dealer frustration, and recalls you can’t surgically contain.
- Traceability Is Reputation Insurance: When every unit’s test data is tied to its serial number, you can isolate problems fast instead of recalling entire batches.
- Ask How Gates Are Enforced, Not Whether They Exist: The revealing question isn’t “do you have quality checkpoints?” — it’s “what physically stops an untested unit from shipping?”
Real process control means quality is built into the production system, not left to human discipline under pressure.
Brand owners and sourcing managers evaluating OEM factory partners will sharpen their due-diligence questions here, preparing them for the detailed evaluation framework that follows.
~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~
The warranty report lands on your desk. Field-failure numbers are climbing — third quarter in a row.
You pull up the factory’s compliance folder. ISO certificate: current. Golden sample approval: signed. Last audit score: passing. Everything looks right on paper. Yet warranty claims keep citing solder joints that look nothing like the approved sample sitting on your shelf.
If this pattern feels familiar, you are not alone. The gap between what a factory demonstrates during evaluation and what actually happens under quota pressure has a name: unenforced QA gates. It is one of the most expensive risks hiding inside commodity OEM partnerships — and you cannot solve it by requesting another compliance deck. The risk sits in whether quality checkpoints are physically enforced by the production system or merely listed in a procedures manual.
Why “Acceptable” OEM Quality Can Still Hide Brand-Level Risk
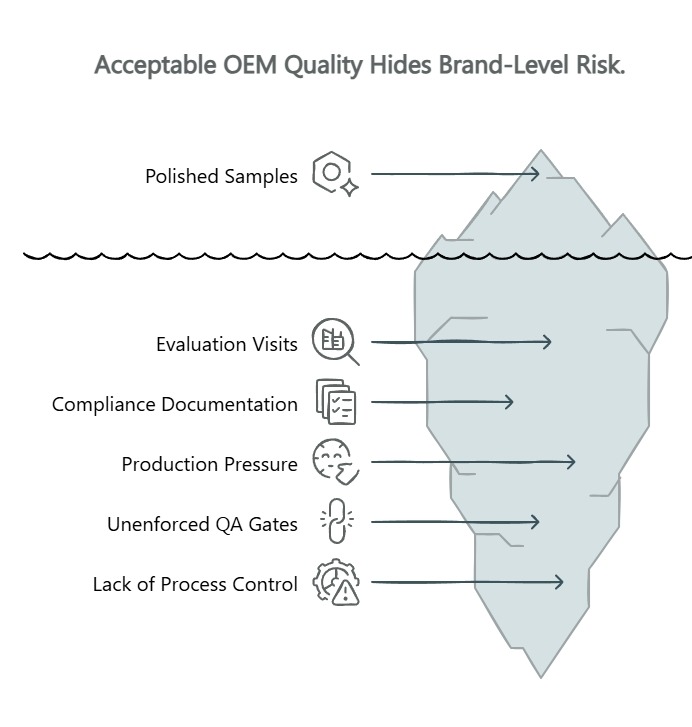
A commodity OEM relationship can look perfectly healthy at the surface. The factory sends polished samples. The compliance documentation checks every box. None of that proves what happens at 4:00 PM on a Thursday when the line is behind on a shipment deadline.
The core mismatch: evaluation visits and golden samples show what a factory can produce. They do not show what the factory will produce — consistently, under pressure, across thousands of units. Your compliance folder may be immaculate while production batches quietly drift out of spec. When QA gates exist as procedural guidelines rather than system-enforced stops, the pressure to meet volume targets will eventually override them.
That distinction separates compliance theater from real process control. Many factories can show forms, inspection stamps, equipment lists, and test stations. Those signals are useful, but they are not sufficient on their own. Real control means the process itself makes it difficult for a nonconforming unit to move forward — not that an operator remembered to follow a checklist. A factory may have IQC, IPQC, FQC, and even a reliability lab on its capability list, but a buyer still needs to know whether those gates are actually enforced in sequence and tied to each unit in a retrievable way.
What Unenforced QA Gates Actually Look Like in the Real World
A QA gate is supposed to be a non-negotiable checkpoint — incoming materials inspected, in-process parameters verified, finished units confirmed to spec before shipping. In a well-governed factory, these gates function like physical barriers. A unit cannot advance until it passes.
In a commodity OEM environment under cost pressure, these gates often become suggestions.
The mechanism is straightforward. When production falls behind schedule, the most time-consuming steps get compressed first. An in-process check that should take 90 seconds per unit gets reduced to a visual glance. A functional test station gets bypassed for a batch because “the first fifty passed.” An incoming material inspection gets skipped because the supplier “has always been reliable.” Each shortcut feels small in isolation.
Consider a production run where function testing or AP testing is inconsistently enforced because the line is pushing to hit an aggressive ship date. A unit that should have been stopped continues downstream. Packaging happens on time. Shipping happens on time. The problem appears later, in the field, when channel partners report unstable performance or an unusual pattern of field failures.
Then comes the conference call where your QA manager asks why Batch 47 measures differently than the approved sample — and the factory representative cannot explain it.
The critical distinction: a documented gate relies on an operator following a procedure. A system-enforced gate — controlled through barcodes, QR codes, or digital routing — physically prevents an untested unit from moving to the next station. As the American Society for Quality notes, quality assurance prevents defects through process design, while quality control catches them after the fact. Unenforced gates collapse that distinction. The difference is between hoping your quality holds and knowing it does.
The Ripple Effect of One Skipped Gate
When an in-process check gets bypassed, untested units mix with tested ones. The line loses the ability to distinguish between verified and unverified output. Spec-drift begins — not as a dramatic failure, but as a quiet erosion of consistency that is invisible inside the factory and expensive outside it.
Hidden spec-drift. Small parameter variations accumulate across runs. Individual units may still pass a loose final check, but the population of shipped products widens beyond the tolerance window your brand promised.
Inconsistent unit performance. Two amplifiers from the same order perform differently in the field. Your dealers notice before your QA team does.
Field failures and warranty exposure. Units that bypassed in-process testing carry defects that surface weeks or months after delivery. Each warranty claim carries direct replacement cost and indirect cost in strained channel relationships.
Channel friction and brand damage. Distributors who receive inconsistent product stop reordering. Trust erodes at the exact point in your distribution chain where it matters most.
Recall risk. A systemic defect from an unenforced gate affects an entire production run. Without unit-level traceability, you cannot isolate which units are affected — forcing a broad recall instead of targeted containment.
That entire sequence starts with one skipped checkpoint. The cost is no longer limited to rework inside the factory. It touches warranty budgets, distributor trust, service workload, and future purchase confidence.
Why Golden Samples and Compliance Checklists Are Not Enough

A golden sample establishes the target — the agreed-upon standard that production must replicate. The problem is not the sample itself. The problem is assuming that approving one proves the factory will replicate it at scale. That sample was likely built with extra attention, possibly by senior technicians, almost certainly without normal production-run time pressure.
A quality management system built around ISO 9001 is not just about checking finished output. It is about building repeatable processes that support consistent results over time. That means golden sample governance must be an ongoing discipline — not a one-time approval that collects dust. The better question during supplier evaluation is not “Can this factory make the sample?” but “What prevents an untested, out-of-tolerance, or wrongly built unit from moving to the next stage when production gets busy?”
Standard compliance checklists carry a similar limitation. A visual AQL inspection catches cosmetic defects — scratches, dents, misaligned labels. It cannot catch invisible acoustic drift: the gradual shift in THD+N, output power consistency, or thermal behavior that occurs when component tolerances stack or a soldering process drifts. These defects generate field failures, and they are fundamentally invisible to visual inspection.
This is where the argument that standard AQL visual inspections are sufficient falls apart. A factory can pass every visual check while shipping units with progressively drifting electrical performance. The real question is whether quality gates are enforced by a system that cannot be overridden by a line supervisor chasing a deadline.
What Real Process Control Looks Like Instead
Real process control is not about adding more inspections. It is about making the inspections that matter impossible to skip.
Route control tied to barcodes or QR codes. Each unit carries a unique identifier tracked through every station. A unit that has not logged a passing result at Station 3 physically cannot be scanned into Station 4. No override. No exception. This is what separates enforced gates from documented ones.
Traceable test data bound to each unit. Every test result — incoming inspection, in-process parametric check, final acoustic measurement — gets linked to that unit’s identifier. If a defect surfaces in the field six months later, the factory can pull the complete production history for that serial number. This capability is the foundation of effective supplier evaluation and recall containment.
IQC, IPQC, and FQC as enforced gates — not labels. These terms appear in nearly every factory’s documentation. The difference is whether they function as active system gates or line items in a manual. In a genuinely controlled environment, IQC prevents uninspected materials from entering production, IPQC catches parameter drift during assembly, and FQC confirms finished-unit compliance before packaging. Each requires logged evidence before the unit advances.
Reliable containment and root-cause capability. When a defect appears, unit-level traceability means the factory can isolate exactly which units are affected and contain the issue without triggering a blind batch recall. This capability alone can separate a manageable quality event from a brand-damaging crisis.
Traceability, as described in GS1’s traceability framework, provides a structured way to track product history, movement, and relevant data across business processes. For an audio brand, the business value is direct: it becomes easier to isolate affected units rather than treating an entire shipment as suspect. Root-cause analysis speeds up because test records are tied to actual units or lots. Supplier conversations become more objective because evidence replaces guesswork. Recall scope narrows when data is strong enough to support targeted containment.
Framing traceability and route control as unnecessary cost misses the point. These systems are reputation insurance. The cost of implementing them is predictable. The cost of not having them becomes visible only when something goes wrong — and by then, the damage is already in your distribution channel.
China Future Sound operates under ISO 9001:2015 and uses ERP and WMS systems with FIFO control. The amplifier production capabilities include SMT, AOI inspection, aging testing, function testing, and AP test stages — each controlled through barcode-based routing that binds test data to individual units. KLIPPEL QC combined with golden sample governance adds acoustic consistency verification. This infrastructure separates a process-governed facility from one relying on operator discipline alone.
Questions to Ask Before You Trust an OEM Partnership
Five questions cut through compliance theater and reveal whether process control is real or ceremonial.
How is movement between test stages enforced?
You want to hear about a digital system — barcodes, QR codes, MES integration — that physically prevents untested units from advancing. If the answer involves operator sign-offs or paper travelers, the gates are not enforced.
Can untested units physically progress through production?
Same question, framed as a stress test. A confident factory will explain exactly how its system blocks untested units. A factory relying on procedural compliance will hesitate.
Is test data bound to individual serial or barcode records?
Unit-level traceability is not optional for serious brand protection. Batch-level recording alone leaves you without containment capability when a defect surfaces downstream.
What evidence shows that IPQC is active, not ceremonial?
Request sample IPQC records from a recent run. Look for logged timestamps, parametric measurements, and pass/fail decisions tied to specific units. Vague or suspiciously uniform records suggest the gate exists on paper only.
How quickly can the supplier isolate affected units if a defect appears?
A factory with genuine traceability answers in hours. A factory without it talks about batch-level recalls and manual investigations.
For a more comprehensive evaluation framework, the factory evaluation checklist provides a structured 30-point assessment, and the OEM audit checklist offers 50 questions designed for pre-contract due diligence.
The Smarter Way to Compare a Lower-Cost OEM Offer
When a competing supplier quotes a lower unit price, the instinct is to compare BOM line items. That comparison is incomplete.
The real comparison is total risk of ownership. A lower-cost offer may save a few percentage points on components. But without system-enforced route control, unit-level traceability, and active in-process quality gates, those savings get consumed by costs that never appear on the initial quote: warranty claims from field failures, dealer confidence eroded by inconsistent units, and recall events that cannot be surgically contained.
Total cost of quality includes everything downstream of the purchase order — the engineering hours investigating field complaints, the warranty replacement costs absorbed by your brand, the channel trust that takes years to build and one bad batch to damage. When those costs are factored in, the “lower-cost” supplier frequently becomes the more expensive one.
The smarter evaluation reframes the question. Instead of “which factory costs less per unit?” ask “which factory can demonstrate that quality is governed by systems rather than intentions?” That shift — from price comparison to process governance — separates brands that scale confidently from brands that scale into warranty exposure.
For a deeper look at how sourcing decisions affect total program cost, why supplier fit beats lowest BOM examines the math. And for teams beginning to question their current relationship, the hidden costs of a commodity OEM partnership maps where the invisible expenses accumulate.
Your Next Step Does Not Have to Be a Supplier Switch
If any of this resonates — if the warranty numbers are trending the wrong direction, or if you suspect your factory’s quality gates are more procedural than enforced — the next step is not a dramatic supplier transition. It is a private assessment.
Start by auditing what you have. Use the five questions above against your current OEM partner. Review warranty and field-failure data for patterns suggesting spec-drift rather than random defects. Compare the compliance documentation in your files against actual production evidence — test records, traceability data, IPQC logs — that your factory can or cannot provide on request.
When reviewing an OEM partner, verify whether IQC, IPQC, FQC, and reliability checks exist as enforced gates with objective pass/fail logic — not just named stages. Confirm whether test-route progression is controlled by barcode, QR, or another system-level method rather than manual sign-off alone. Check that parametric test data is bound to each specific unit or serial identifier to guarantee true traceability. Assess whether FIFO discipline is enforced through ERP and WMS or handled informally. And determine whether golden sample management is maintained as an active, ongoing discipline rather than a one-time approval.
Those checks do not require a full supplier transition. They are a disciplined first filter that separates factories that look organized from factories that can actually protect a product line at scale.
The 72-hour OEM audit guide offers a structured approach to evaluating quality gates in a compressed timeframe, designed for a decision-maker who needs clarity before committing to a larger evaluation. For deeper validation, the supplier selection framework provides a broader lens.
Unenforced QA gates do not announce themselves. They hide behind polished samples and passing compliance scores until the cost shows up in your warranty line, your dealer relationships, and your brand reputation. The brands that avoid this trap are not the ones with the biggest budgets. They are the ones who learned to ask the right questions before signing.
Clear gates. Controlled flow. Traceable units. If the route is not controlled, the risk is not controlled.
Our Editorial Process
Our expert team uses AI tools to help organize and structure our initial drafts. Every piece is then extensively rewritten, fact-checked, and enriched with first-hand insights and experiences by expert humans on our Insights Team to ensure accuracy and clarity.
About the China Future Sound Insights Team
The China Future Sound Insights Team is our dedicated engine for synthesizing complex topics into clear, helpful guides. While our content is thoroughly reviewed for clarity and accuracy, it is for informational purposes and should not replace professional advice.






